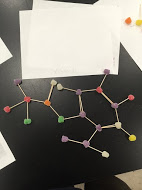
Today in class, we learned about the molecular structure of atoms. To display our knowledge, we made molecular structures of atoms using gumdrops, toothpicks, and raisins. We compared the structures of inorganic atoms and organic molecules. We were able to see that organic molecular structures were a little more complex. I learned how to make dot structures out of a given chemical formula. During the lab we also studied covalent bonds and how to atoms that have bonded together are structured while still following the octet rule. Below are pictures of the molecular structure out of gumdrops. The corresponding names for the structures are labeled near them. There is a key on each paper explaining what each color of gumdrop represents.
|
How was your experience this semester?
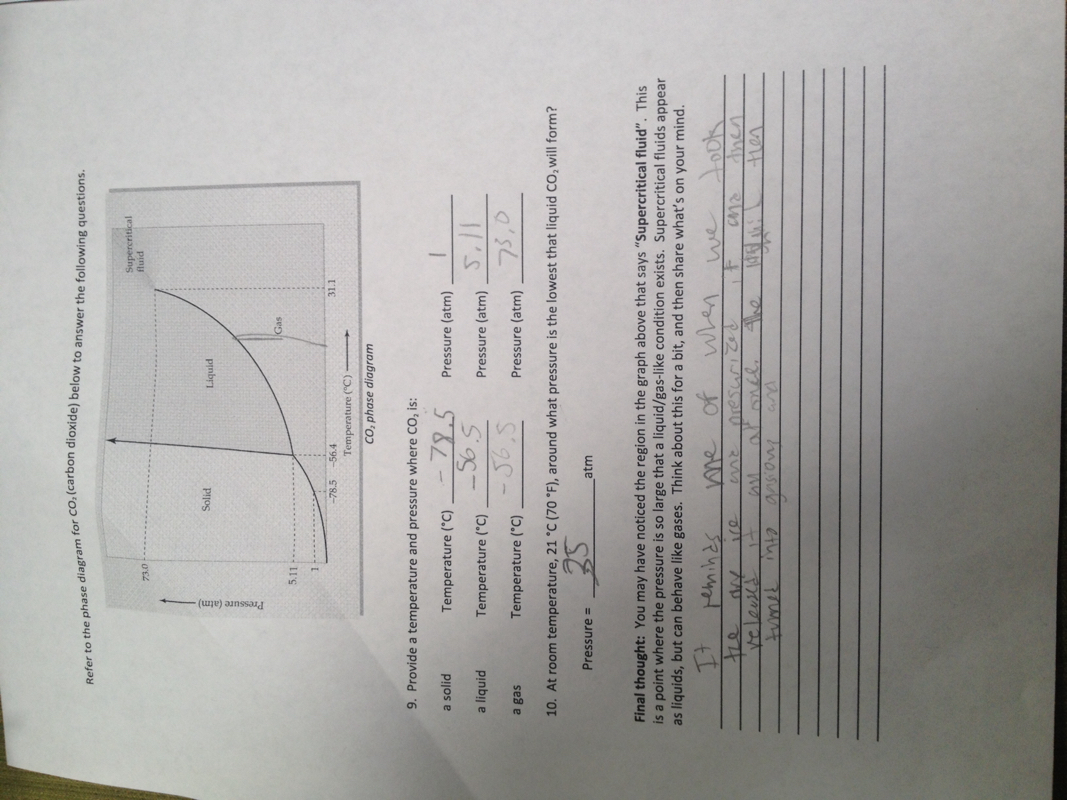
This semester in Andrews class was great. The difference in the amount of choice and freedom we had in this class compared to my other classes made me look forward to 4th and 5th period. I hate coming to school because I feel like I am just wasting my time learning about crap that I don't need to know. The only time were I feel like I am actually learning is in Andrews class. The stuff we learn in Andrews isn't memorized. It's deeper learning that is driven by your eagerness to pursue your interests. The only reason I want to do well in my other classes is for the grade. In Andrews, I want to exceed my own expectations of what I can do and the grade isn't always nagging me. In other classes I'm trying hard not to fall asleep but in chemistry I'm wide awake. What did you learn about yourself as a learner? I learned that to really reach my potential as a learner, I need to have the freedom to explore education my way. Not just jump though hoops as many of my other classes have had me do in the past. I am most engaged when my learning is driven through my own interests. I realize that my learning won't always be driven by my interests but every once in a while it would be nice to be some choice. When were you motivated by curiosity? When weren't you? I can see a pattern where towards the beginning of each section, my work is more creative and I think this because I have no worries about grades in the begging. As each section comes to a close, my work starts to become mostly worksheets to meet the deadline (which I don't mind.) What will you take away from this class? I will take away the appreciation for choice in learning. This semester pretty much restored my faith in the 10th grade because it was all just filled with too much directions and not enough of me being able to take the reins of my learning and steer it towards my curiosity and my passions. This semester was really awesome and I hope that I will be able to keep pursuing my curiosity through education.  This was my very last rocket for this semester in chemistry. My partner Carlos and I made this rocket from scratch. The engine and all. It was a really cool way to end off our rocket building careers for the year. I would have liked to see more success at the launch but the experience of building the rocket with my friend was worth it. The rocket had a decent burn time but it didn't have enough thrust to get off the launch-pad. It would have been better if we didn't have as much weight on it. The nosecone was especially heavy because of the way we made it using cardboard tubes gradually decreasing in size. We couldn't find the balance of light and heavy because if it was too light the rocket would tip to the side and with too much weight it wouldn't get off the pad. For the engine we used a mix of sorbitol and potassium nitrate in a 36 to 64 percent ratio. We melted it down and poured it into the motor casing and let it harden overnight. Then we drilled a hole in the bottom to funnel out all of the energy that is being combusted through the bottom of the rocket. Fortunately for us, if Carlos and I wish to pursue rocketry even further looking forward we can always walk down to Andrews class and take the next rocketry project head on. So, I don't really want to say that it ends here because it doesn't have to. I think the Fancy Turtle deserves the a successful launch. I would be open to working with Carlos again but if possible I would like to have some other people on the team to help us make the gnarliest rocket ever. Now that I have the skills to make a rocket from scratch including the engine, future rocket projects will be much easier and I can help others as well. Making your own rockets and engines takes lots of time and patience. Being able to work well in the partnership was important too. Carlos and I had the same vision for the fancy turtle in the start but both of us were open to each others ideas for improvement . In this lab, my partner Garrett and I made a small rocket engine using table sugar and potassium nitrate. We covered a lot during the lab regarding chemical reactions and I felt like making the small engine helped me visualize what I was learning on the board. The goal of the lab was to take a little amount of solid, and turn it into a lot of gas. We did this by taking the glucose, fructose (table sugar) and the potassium nitrate and adding energy to it. By doing this, we created an exothermic reaction, which gave off a lot of heat and energy pushing out motor into the sky. The solids turned into gasses, which takes up more space (as seen in the hydrogen lab) and we were left with, Potassium carbonate, Co2, and chemical nitrate pushing themselves straight out of the bottom giving us lift. We measured out our substances using stoichiometry which was basically transferring moles to grams. For the potassium nitrate, the amount to use we were given was, 48kno3 and we needed to transfer that into grams. What we then did was take the atomic weight of each element on there, 31K, 14N, 16O3 so 48O = 93. Then we multiplied that by 48 to get 4,464g. If we repeat the process with the table sugar, we end up with 1710g. Instead of having to use so many grams to make a small rocket, we added the two numbers together and found the percentage of each substance. 1710+4464= 6174 / 1710= 36% sugar and 6174/4464= 74% potassium nitrate. Reflection This experiment was a great success for my partner Garrett and I. Our rocket was one of the highest ones to go off. It even almost set the school on fire! But we can save that for another day. To get better results, I think we could have packed more fuel into the rocket. The rocket had great initial lift but burned out pretty quick after it left the ground. I am definitely thinking about how I can use this information I learned for my next rocket build. I will need to make my own motor from scratch and this has given me some good ideas about what methods I can pursue. |
AuthorWeWrite something about yourself. No need to be fancy, just an overview. Archives
May 2015
Categories |








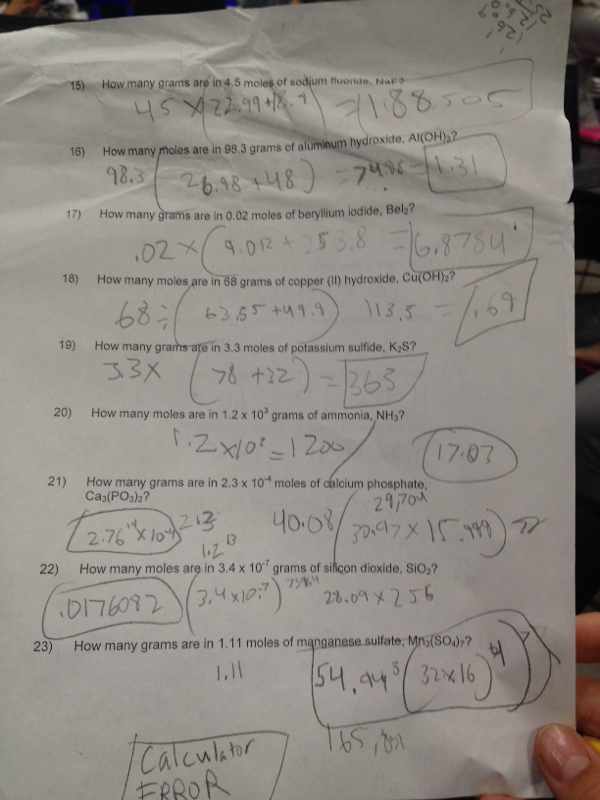





 RSS Feed
RSS Feed
